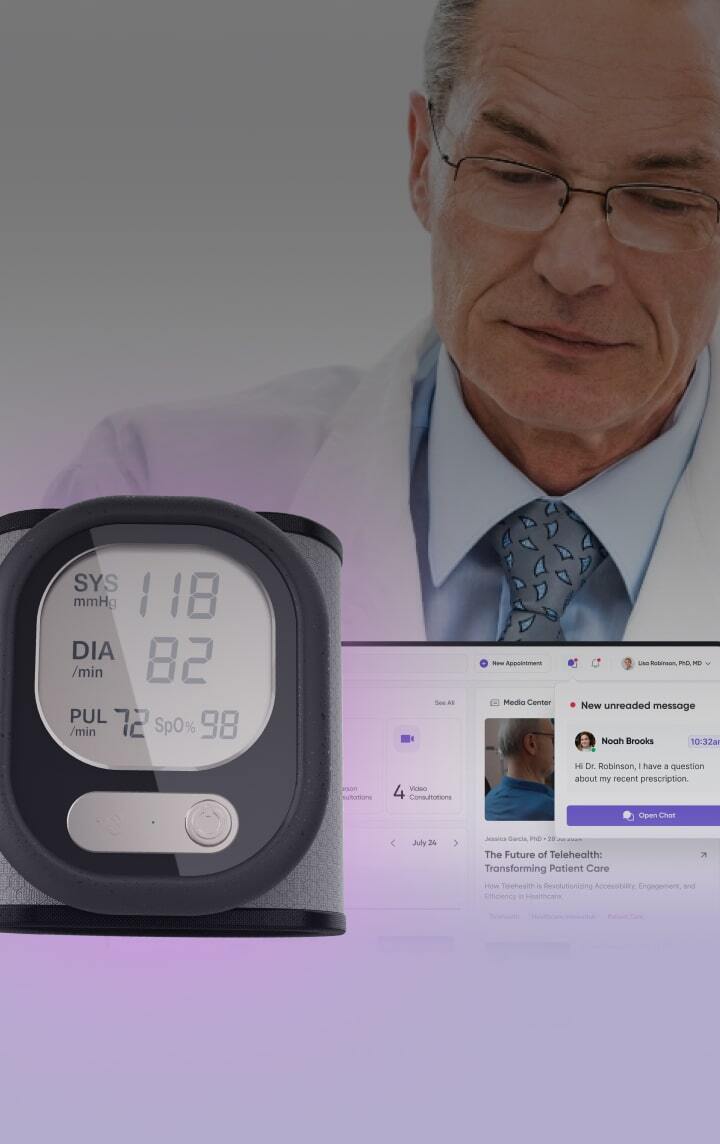
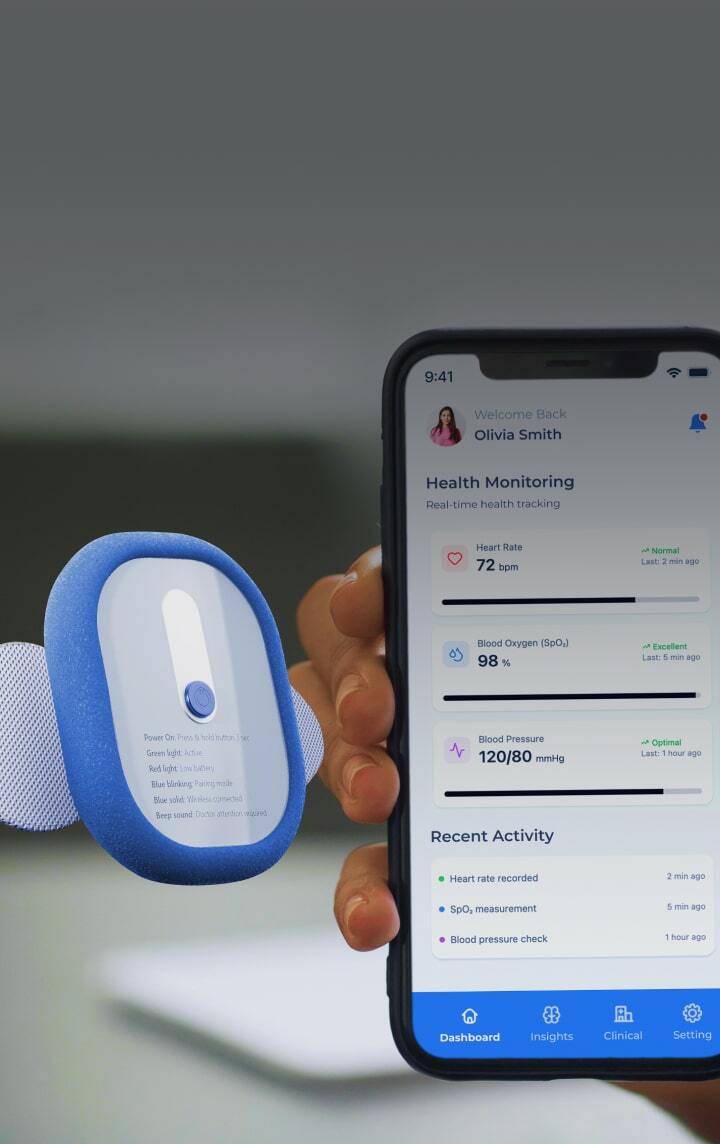
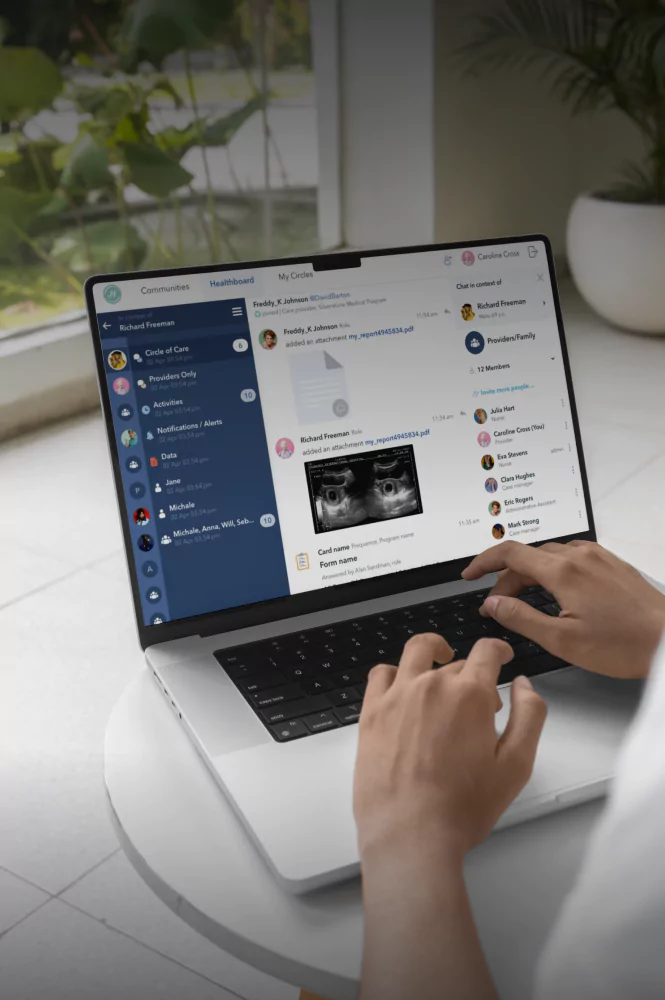
Healthcare product design & development services
Full-cycle engineering for medical devices, SaMD, HealthTech and smart hospitals. We bridge the gap between R&D and manufacturing, ensuring compliance, security, and scalability from day one.
Years in healthcare
Built-in compliance
FDA & CE approvals
Experts
Certified QMS
Healthcare product engineering and development services
Technology stack
-
Rust
-
C
-
C++
-
Kotlin
-
Bootloader
-
Linux Kernel
-
AWS IoT
-
Arduino
-
ESP32
-
STM32
-
NRF52
-
Zephyr
-
LoRaWAN
-
MQTT
Why partner with Yalantis
-
Proven track record in regulated healthcare
We deliver software for Class II & III devices and complex digital health platforms, leveraging 15+ years of experience to handle safety-critical environments where failure is not an option.
-
Advanced technology & Rust expertise
From memory-safe Rust for firmware to scalable cloud architectures. We ensure 99.99% uptime and solve high-load challenges that generalist dev shops cannot handle.
-
Specialized healthcare engineering teams
Scale your R&D in 2-4 weeks with engineers who understand clinical workflows. We ensure critical domain knowledge remains consistent throughout the lifecycle.
-
Audit-ready security & compliance
An ISO 13485 and ISO 9001 certified partner. We align your product with FDA, MDR, HIPAA, and GDPR standards from day one to streamline certification and audits.
-
End-to-end ecosystem synergy
We bridge hardware, cloud, and clinical systems. Unifying low-level firmware, patient apps, and EHR integration into a seamless, interoperable healthcare ecosystem.
-
Engineered for business growth
Maximize value by optimizing device BOMs and software TCO. We ensure your product makes business sense, whether it’s a mass-produced device or a scalable SaaS platform.
What our clients say
Our collaboration and engagement models
-
Time and materials
– Long-term project and dynamic requirements
– Scope of work is not fully specified
– May need to alter the scope and the workload
-
Fixed price
– Clear requirements and strict deadlines
– Project with a limited scope of work
– We manage the entire product development lifecycle.
-
Managed team
– Yalantis provides professionals (or a whole team) with the required expertise
– We take responsibility for our specialists’ performance
– Specialists follow your corporate values and development processes -
Managed product
– Yalantis holds a wide range of responsibility, designing and co-creating a product with you
– We provide the development team for your project
– We manage the entire product development life cycle -
Managed service
– Yalantis helps you with just one or a number of services, such as design, development, QA, and DevOps
– We provide the required experts (or team of experts)
– We fully manage the entire development process
How we work on your compliant healthcare solution
-
Strategy
Some clients need full development, others only regulatory support or consulting. We begin by clarifying what’s needed and what results matter most to you.
-
Planning
Our team prepares a roadmap that covers everything from regulatory requirements to technical design. This gives clarity on tasks, responsibilities, and timelines.
-
Execution
The agreed services are delivered by our experts. You can engage us for a single stage or involve us across the entire process.
-
Support
The engagement is finalized by transferring results into practice. Ongoing support is available whenever it’s needed.
Healthcare product design & development insights

How Connected Healthcare Devices Are Reshaping Care Delivery and Driving Market Growth
Learn what connected healthcare is, it’s fundamental difference from telehealth, opportunities it opens for businesses, and what it takes to develop a connected healthcare device.

HIPAA Compliance Checklist for Healthcare Software Development
This guide gives comprehensive information on how to ensure HIPAA compliance. You’ll also learn how to implement safeguards to meet the HIPAA Security Rule.

Rust for Medical Devices: Certified Software for Safety-Critical Systems
Explore how Rust can enhance medical device software with memory safety, performance, and reliability, helping developers build secure and dependable embedded systems.
FAQ
How do you ensure FDA, CE, and HIPAA compliance during development?
Compliance is built into every stage of our work. Our team follows established standards, prepares the required documentation, and manages submissions directly with regulators. With our product engineering services in healthcare, you can be assured that your product is developed in line with safety, quality, and data protection requirements.
What makes your healthcare product engineering services different from other providers?
Our healthcare product development company has over 15 years of experience in product engineering for healthcare. We’ve supported more than 50 FDA and CE submissions and keep a 90% first-pass approval rate. Unlike many providers, we bring together regulatory consultants and engineers, so clients get both compliance and development expertise in one place. Whether it’s medical device design, SaMD, or modernizing healthcare systems, we deliver end-to-end solutions that are fully compliant and ready for the market.
How much time does it usually take to bring a healthcare product to market with your support?
Timelines vary depending on the scope. A new medical device or SaMD can take 12–24 months to develop and certify, while shorter projects like compliance consulting or system integration may be completed in weeks or months. You can always reach our consultancy team to discuss your project and get a realistic timeframe.
Can I hire you just for one service, like regulatory consulting or EHR integration?
Yes, our healthcare product development services are flexible. Some clients engage us for full projects, while others need help with a specific stage such as compliance, platform integration, or EHR modernization. We adapt the team and scope to match your priorities
What engagement models do you offer, and how flexible are they?
We adapt to different types of projects. Some clients work with us on a fixed price basis when the scope is clear. Others choose time and materials when requirements may change. We also provide managed teams that follow your processes, or take full ownership through managed product and managed service models. However you prefer to work, our healthcare product engineering solutions can scale to fit.
How many domain experts do you have in your healthcare product development company?
Our agency brings together more than 200 engineers, designers, and regulatory specialists with direct experience in healthcare product engineering. Depending on your needs, we form dedicated teams that can handle full-cycle projects or focus on specific tasks like integration or reengineering existing healthtech solutions.
Contact us

got it!
Keep an eye on your inbox. We’ll be in touch shortly
Meanwhile, you can explore our hottest case studies and read
client feedback on Clutch.

Nick Orlov
IoT advises
How to get started with IoT development
-
Get on a call with our Internet of Things product design experts.
-
Tell us about your current challenges and ideas.
-
We’ll prepare a detailed estimate and a business offer.
-
If everything works for you, we start achieving your goals!