Digital Labeling Services
for Pharma
A validated, end-to-end service to eliminate paper-based risks and create a fully digital, GxP-compliant labeling process. We are a technical partner for Heads of Quality, Operations, and Clinical Supply frustrated with the constant deviations, manual errors, and compliance risks of traditional paper labels.
Challenges Our Digital Displays Solve
-
Costly deviations and CAPAs from manual label errors
Instead of managing a stream of deviations from incorrect printing, application, or transcription errors, our service implements a fully system-controlled process. This eliminates the human error inherent in manual, paper-based workflows and the associated risk to patient safety.
-
Risky and cumbersome manual relabeling for updates
Forget the operational nightmare of recalling products to a depot for manual relabeling just to update an expiry date. Our service validates a digital process that allows you to update label content instantly and remotely, reducing relabeling lead time from months to a single day.
-
Product loss from physically damaged or illegible labels
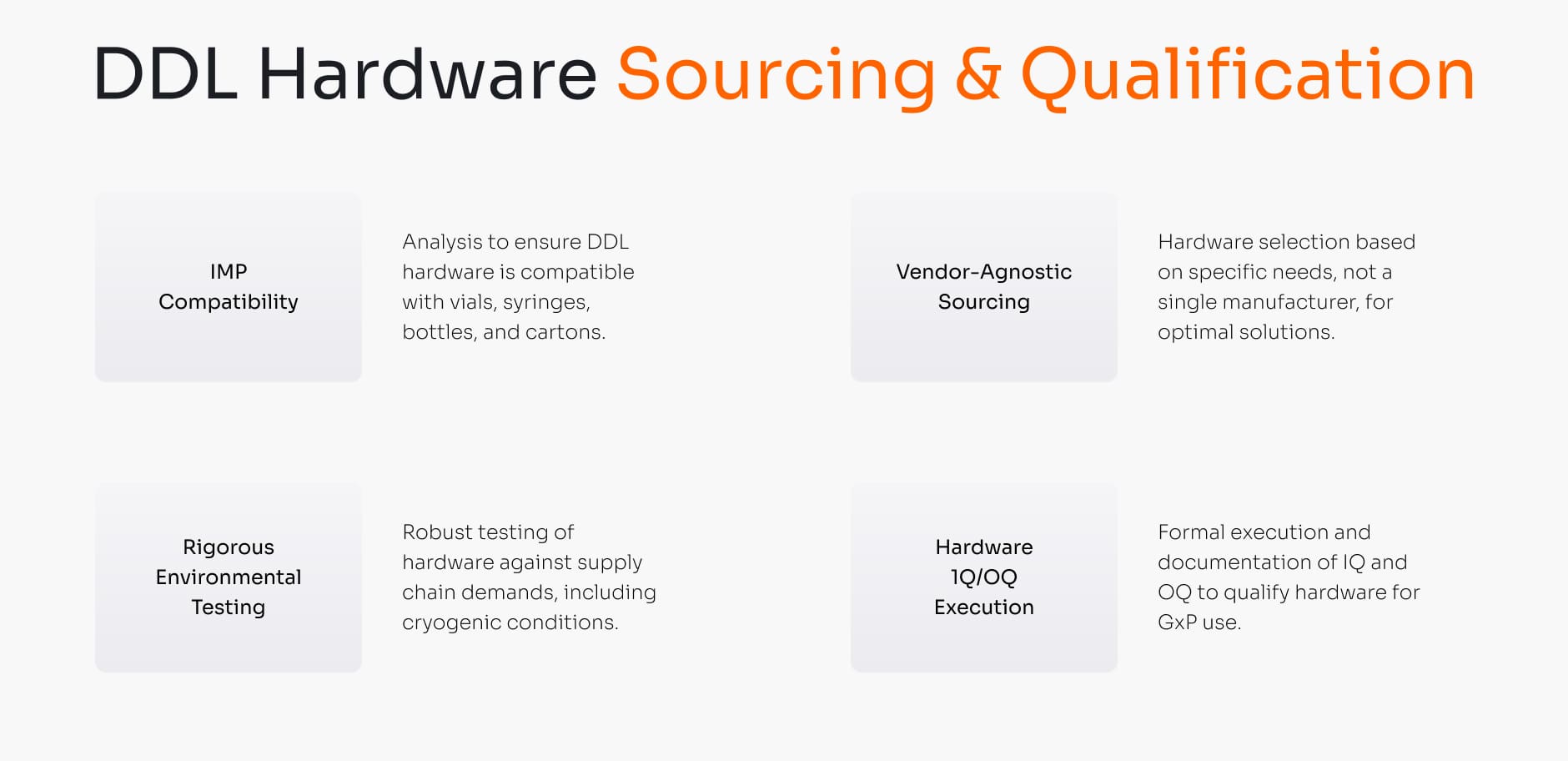
Paper labels are easily compromised by moisture, cryogenic storage, or simple abrasion, leading to faded ink, flaking, or labels falling off entirely. This can render a high-value IMP unsaleable. We qualify hardware designed to withstand the rigors of the clinical supply chain.
-
Lack of GxP-compliant traceability for label changes
Manual relabeling processes are not inherently traceable, creating a significant compliance gap and risk during audits. Our validated, 21 CFR Part 11 compliant system provides an unalterable audit trail for every single label update, ensuring you are audit-proof from day one.
-
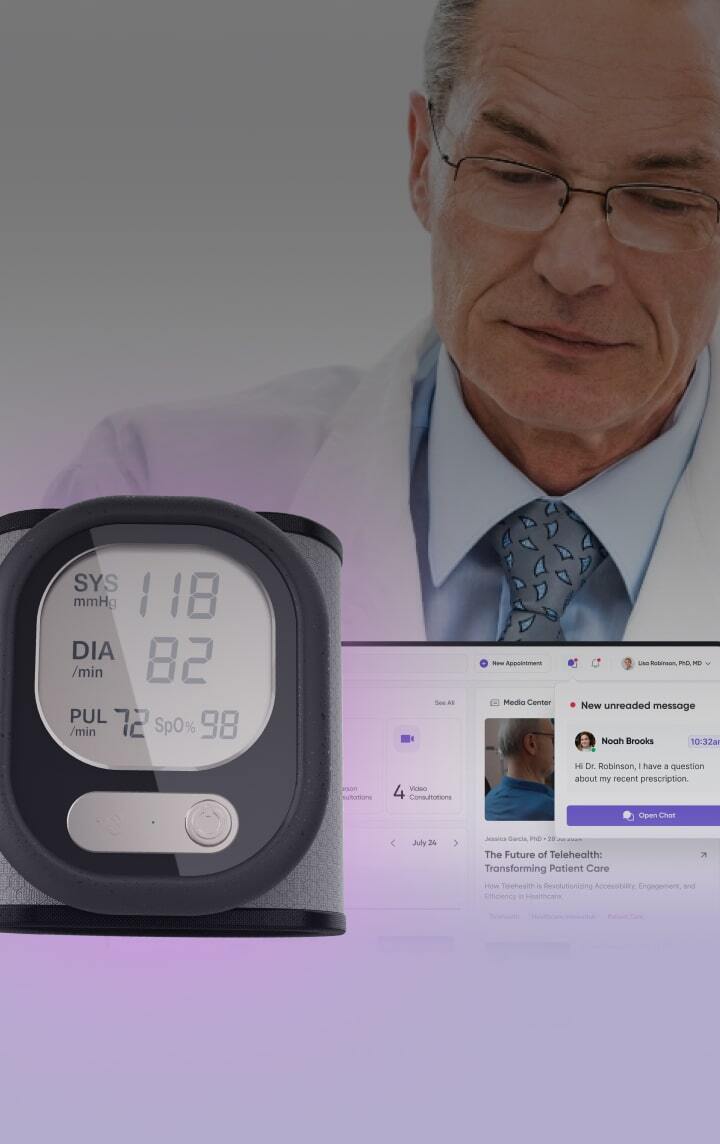
Illegible text and complex, multi-language booklet labels
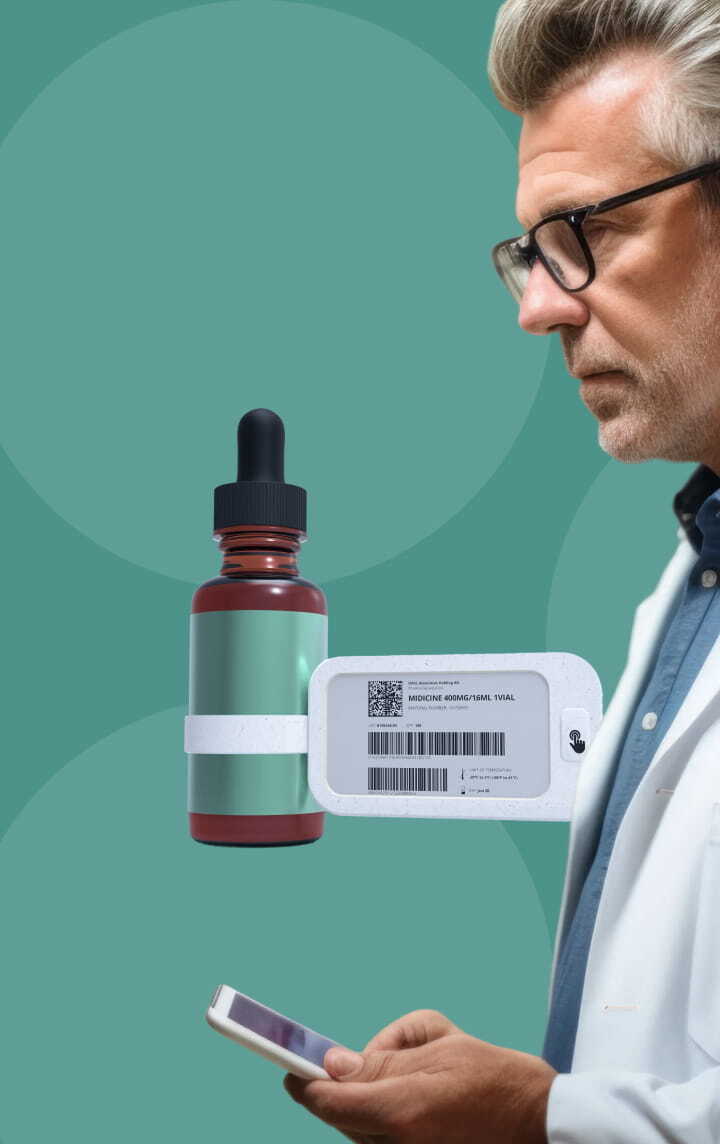
Our DDL solution offers a clear e-paper screen, eliminating the need to decipher small fonts on vials or lengthy booklets. This technology provides adjustable font sizes and easy page-flipping, ensuring critical information is always legible and simple to navigate.
-
Inflexible inventory and high waste from static, protocol-specific labels
Paper labels are static and must be pre-printed for specific protocols and countries, leading to long lead times and high waste. Our DDL platform lets you use a single, protocol-agnostic batch and update the label digitally just before dispensing, slashing both waste and lead times.
Yalantis Digital Display Labeling Services
Our success stories
Benefits of a Validated Digital Display Labeling
-
Drastically Reduce Labeling Deviations
By replacing manual, error-prone paper processes with a system-controlled, validated workflow, you eliminate the primary source of label-related deviations, rework, and CAPAs.
-
Maximize Operational & Supply Chain Flexibility
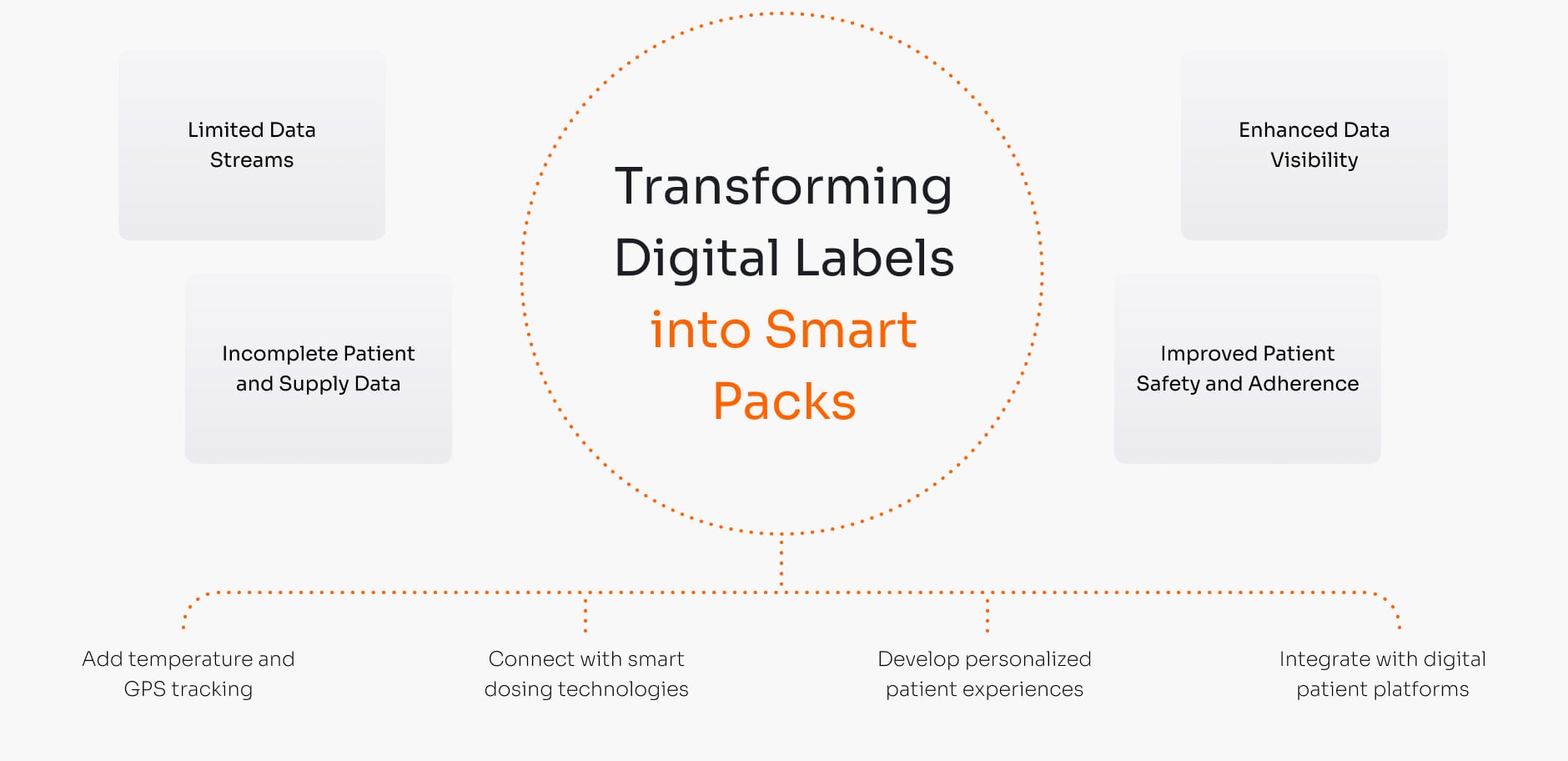
Instantly update expiry dates, add new languages for different countries, or re-assign pooled inventory to different trials. This agility reduces product waste and shortens critical lead times, making your supply chain more resilient.
-
Create a Modern, Defensible, and Compliant Supply Chain
Our service delivers exactly what auditors want to see: a modern, 21 CFR Part 11 compliant system. You move away from untraceable manual steps to a robust process that provides unalterable evidence of your label’s content at every point in its journey.
-
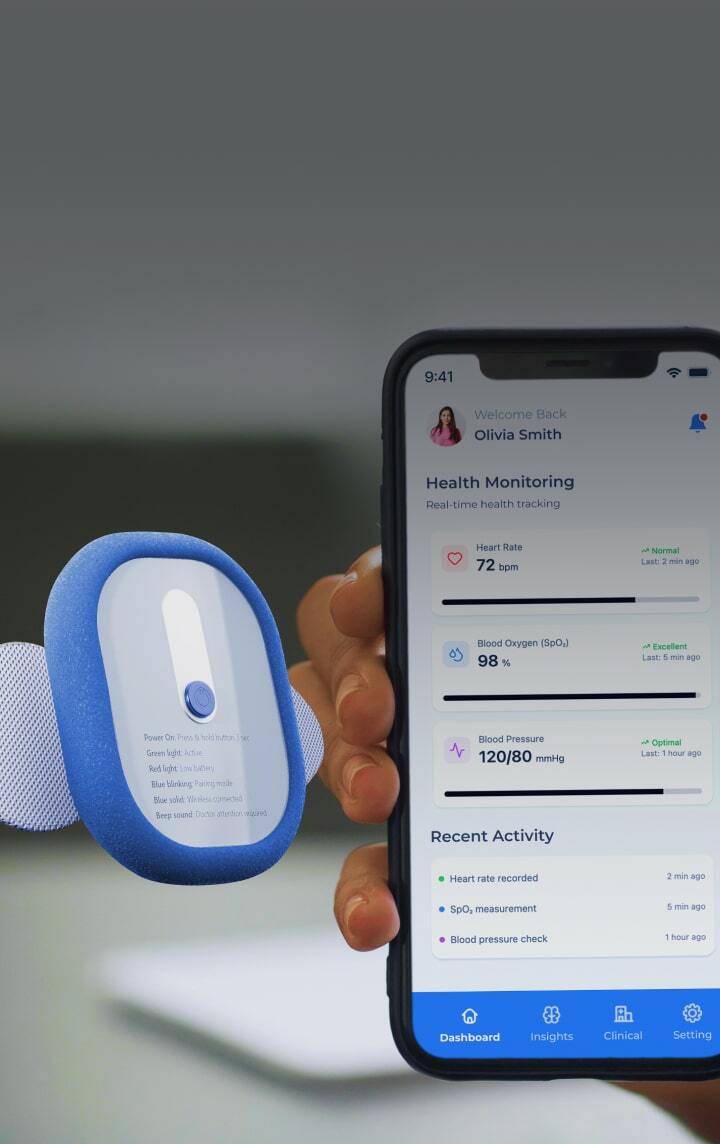
Enhance Patient Safety and Site Experience
DDLs provide clear, legible information, with the ability to adjust font size or display information in a patient’s preferred language. This improves the user experience, reduces the administrative burden on clinical site staff, and enhances overall patient safety and adherence.
-
Streamline Initial Label Creation and Production
Our service streamlines initial label creation and production by replacing the labor-intensive process of designing, printing, and delivering complex paper booklet labels with an agile, digital workflow, significantly reducing the timeline and effort for initial label production.
-
Enterprise-Grade Security & Scalability
Our validated process streamlines labeling and distribution, eliminating paper’s long lead times and expediting supply operations. This reduces supply delays and shortages, contributing to faster drug development and quicker patient access to life-saving treatments.
Testimonials from our clients
FAQ
What is the difference between your service and just buying DDL hardware?
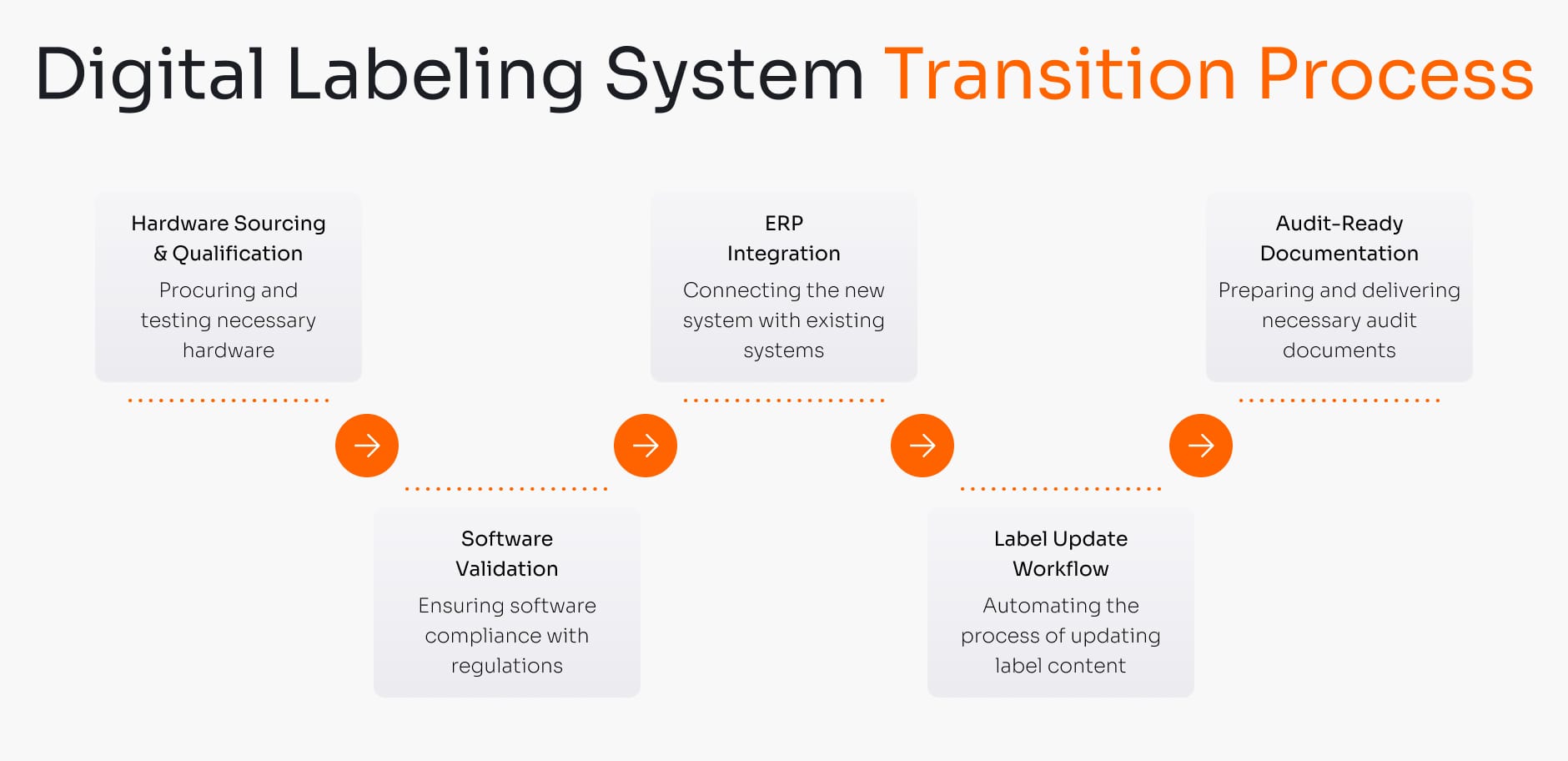
Hardware is just a component; a validated process is the solution. Our service is the critical GxP bridge. We manage the entire validation lifecycle (IQ/OQ/PQ) and ensure the end-to-end software process, from your ERP to the DDL system, is 21 CFR Part 11 compliant. Buying hardware leaves the entire integration, validation, and compliance burden on your internal team.
How do DDLs handle ultra-low temperature storage?
This is a known challenge for electronics. Our hardware selection process is critical here. While most e-paper displays cannot be updated in frozen conditions, the information remains perfectly legible. The update (e.g., via NFC) is performed at a qualified point in the process, such as during packing or just before dispensing, in a system-controlled and validated manner.
How do you validate the “right” update goes to the “right” label?
This is the core of our software validation. The system maps unique identifiers. Each DDL has a unique, unchangeable ID (like an NFC UID). The update order from your ERP is linked to this specific ID. The system requires a “positive encoding confirmation” to verify that the DDL was updated correctly, creating a closed-loop, documented process.
What about sustainability? Aren’t electronics more wasteful than paper?
While DDLs involve electronics, the waste from traditional labeling is significant, including label re-work, wasted inventory from label errors, and entire shipments being discarded. By enabling inventory pooling and eliminating re-work, DDLs can dramatically reduce overall product waste. Our service also focuses on sourcing reusable DDLs to create a circular model, further improving the sustainability case.
Are health authorities like the FDA & EMA actually accepting digital labels instead of paper?
Yes, because unlike past “e-label” concepts that relied on QR codes, our DDL solution displays all required regulatory content as human-readable text directly on the label itself. Health authority concerns were focused on solutions where the required text was not visible on the product. Our fully validated, 21 CFR Part 11 compliant process ensures the method of displaying and updating this digital text is secure, traceable, and defensible.
Contact us

got it!
Keep an eye on your inbox. We’ll be in touch shortly
Meanwhile, you can explore our hottest case studies and read
client feedback on Clutch.

Nick Orlov
IoT advisor
How to get started with IoT development
-
Get on a call with our Internet of Things product design experts.
-
Tell us about your current challenges and ideas.
-
We’ll prepare a detailed estimate and a business offer.
-
If everything works for you, we start achieving your goals!